Back
Introduction: Cohort B of the phase 2 KEYNOTE-057 (NCT02625961) study evaluated pts with BCG-unresponsive HR NMIBC with papillary tumors (no carcinoma in situ [C-IS]) who were ineligible for or declined radical cystectomy (RC). Herein we present the primary efficacy results and outcomes of pts who underwent subsequent cystectomy.
Methods: Pts aged =18 y with BCG-unresponsive HR NMIBC with papillary tumors only (high-grade Ta or any-grade T1) at baseline and ECOG PS score of 0-2 received pembro 200 mg every 3 wk for =35 cycles (~2 y). Primary end points for cohort B were 12-mo disease-free survival (DFS) rate of HR NMIBC by central pathology/radiology review and safety. Secondary efficacy end points included 12-mo DFS rate of any disease (low-grade Ta, HR disease, and progressive disease). Time to RC and pathologic staging at time of RC were evaluated.
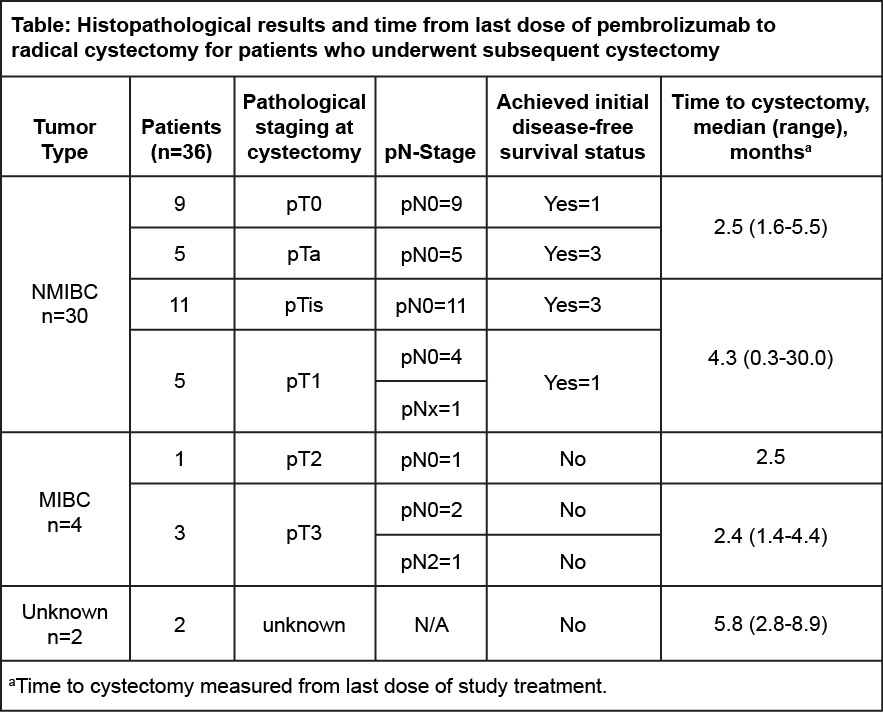
Results: A total of 132 pts were enrolled with a median (range) of 10.0 (6.0-33.0) prior BCG instillations; 57 pts (43.2%) had T1 disease, and 75 pts (56.8%) had high-grade Ta disease. Pts received a median (range) of 9.5 cycles (1.0-35.0) of pembro. Median (range) follow-up was 45.4 mo (14.9-77.1). The 12-mo DFS rate of HR NMIBC was 43.5% (95% CI, 34.9-51.9); 12-mo DFS rate of any disease was 41.7% (33.1-50.0). Treatment-related AEs occurred in 97 pts (73.5%); 19 pts (14.4%) had a grade 3/4 treatment-related AE. No deaths from treatment-related AEs occurred. A total of 36 pts (28.3%) underwent RC after stopping pembro; 8 pts (18.6%) achieved initial disease-free status and 28 (45.9%) did not. Median (range) time from last dose of pembro to RC was 6.5 mo (1.0-16.0) for those who achieved initial DFS status and 2.8 mo (0.3-30.0) for those who did not. Pathologic staging at RC is presented (table). Of 36 pts who underwent RC, 4 known pts (11.1%) had upstaging to MIBC; none of these pts experienced an initial disease-free status.
Conclusions: After approximately 45 mo of follow-up, pembro monotherapy demonstrated antitumor activity in pts with BCG-unresponsive non-CIS papillary NMIBC. Rates of upstaging at time of RC are consistent with previous reports, suggesting that the window of opportunity for subsequent curative RC is generally preserved. SOURCE OF
Funding: Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA
Podium Session
Session: LBA03: Late-Breaking Abstracts III - Cancer
LBA03-08: Pembrolizumab (pembro) for Patients (pts) With High-Risk Non-Muscle-Invasive Bladder Cancer (HR NMIBC) Unresponsive to Bacillus Calmette-Guerin (BCG): Efficacy and Evaluation of Subsequent Cystectomy From Cohort B of the Phase 2 KEYNOTE-057 Study
Sunday, April 30, 2023
4:50 PM – 5:00 PM CST
Location: S404A

Eric A. Singer, MD, MA, FACS
Rutgers Cancer Institute of New Jersey
Podium Presenter(s)
Introduction: Cohort B of the phase 2 KEYNOTE-057 (NCT02625961) study evaluated pts with BCG-unresponsive HR NMIBC with papillary tumors (no carcinoma in situ [C-IS]) who were ineligible for or declined radical cystectomy (RC). Herein we present the primary efficacy results and outcomes of pts who underwent subsequent cystectomy.
Methods: Pts aged =18 y with BCG-unresponsive HR NMIBC with papillary tumors only (high-grade Ta or any-grade T1) at baseline and ECOG PS score of 0-2 received pembro 200 mg every 3 wk for =35 cycles (~2 y). Primary end points for cohort B were 12-mo disease-free survival (DFS) rate of HR NMIBC by central pathology/radiology review and safety. Secondary efficacy end points included 12-mo DFS rate of any disease (low-grade Ta, HR disease, and progressive disease). Time to RC and pathologic staging at time of RC were evaluated.
Results: A total of 132 pts were enrolled with a median (range) of 10.0 (6.0-33.0) prior BCG instillations; 57 pts (43.2%) had T1 disease, and 75 pts (56.8%) had high-grade Ta disease. Pts received a median (range) of 9.5 cycles (1.0-35.0) of pembro. Median (range) follow-up was 45.4 mo (14.9-77.1). The 12-mo DFS rate of HR NMIBC was 43.5% (95% CI, 34.9-51.9); 12-mo DFS rate of any disease was 41.7% (33.1-50.0). Treatment-related AEs occurred in 97 pts (73.5%); 19 pts (14.4%) had a grade 3/4 treatment-related AE. No deaths from treatment-related AEs occurred. A total of 36 pts (28.3%) underwent RC after stopping pembro; 8 pts (18.6%) achieved initial disease-free status and 28 (45.9%) did not. Median (range) time from last dose of pembro to RC was 6.5 mo (1.0-16.0) for those who achieved initial DFS status and 2.8 mo (0.3-30.0) for those who did not. Pathologic staging at RC is presented (table). Of 36 pts who underwent RC, 4 known pts (11.1%) had upstaging to MIBC; none of these pts experienced an initial disease-free status.
Conclusions: After approximately 45 mo of follow-up, pembro monotherapy demonstrated antitumor activity in pts with BCG-unresponsive non-CIS papillary NMIBC. Rates of upstaging at time of RC are consistent with previous reports, suggesting that the window of opportunity for subsequent curative RC is generally preserved. SOURCE OF
Funding: Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA

