Back
Introduction: Fumarate hydratase-deficient renal cell carcinoma (FH-RCC) is a rare and highly aggressive cancer mainly caused by germline or somatic aberrant of FH gene. Unfortunately, there are no standard treatment. Here we report the preliminary results of a phase II study investigating the efficacy and safety of sintilimab in combination with axitinib in patients with FH-RCC.
Methods: Patients were treated at mutli-center in hospitals of the republic of China. Eligibility criteria included age = 18 years and newly diagnosed as FH-RCC by FH immunohistochemistry and next-generation sequencing or multiplex ligation-dependent probe amplification. Patients received sintilimab (intravenous injection, every 3 week) in combination with axitinib (5mg, orally taken per day) as first-line treatment until disease progression or intolerant to treatment. The primary end point was objective response rate (ORR; RECIST v1.1). This study is registered with ClinicalTrials.gov, NCT04387500.
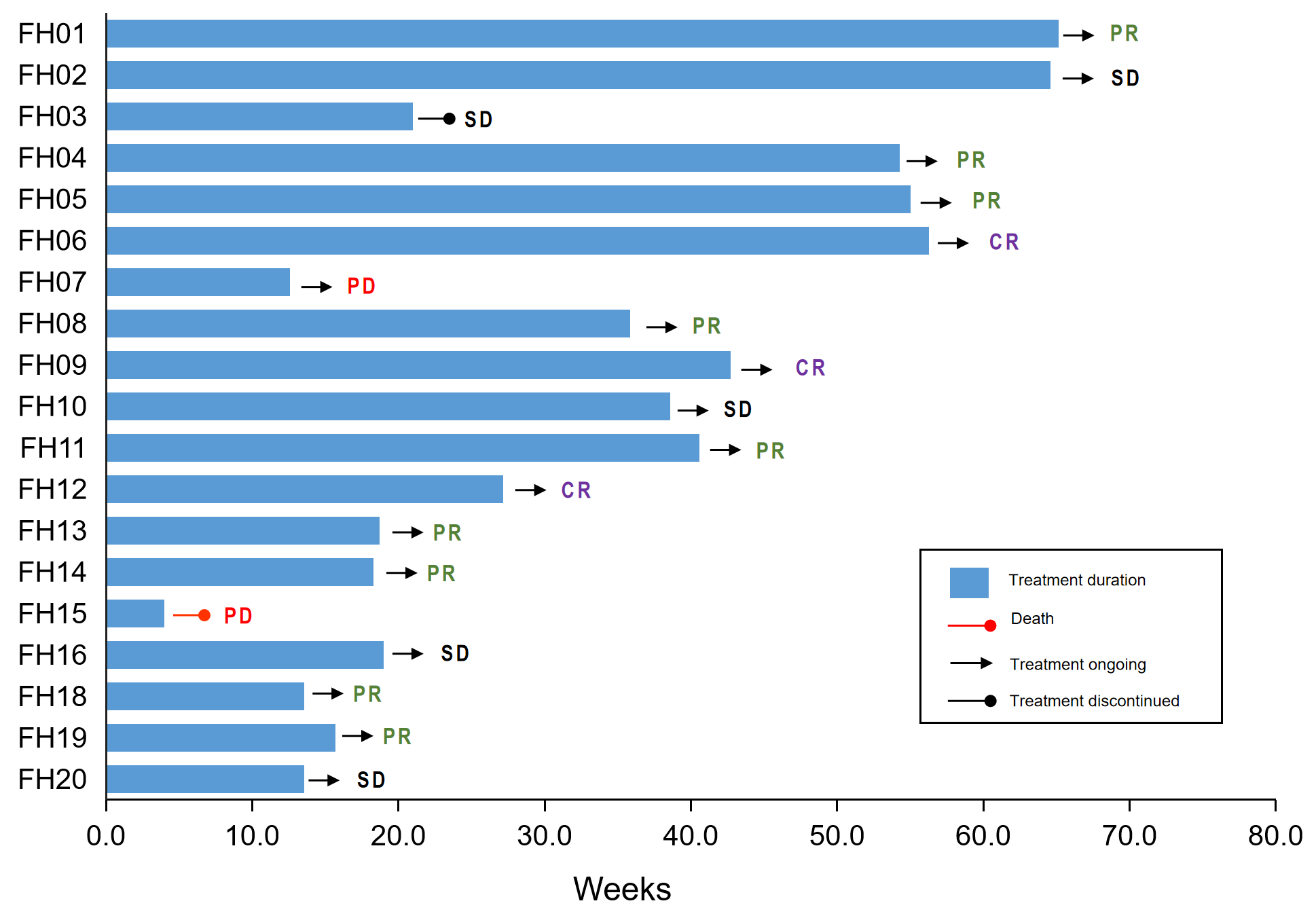
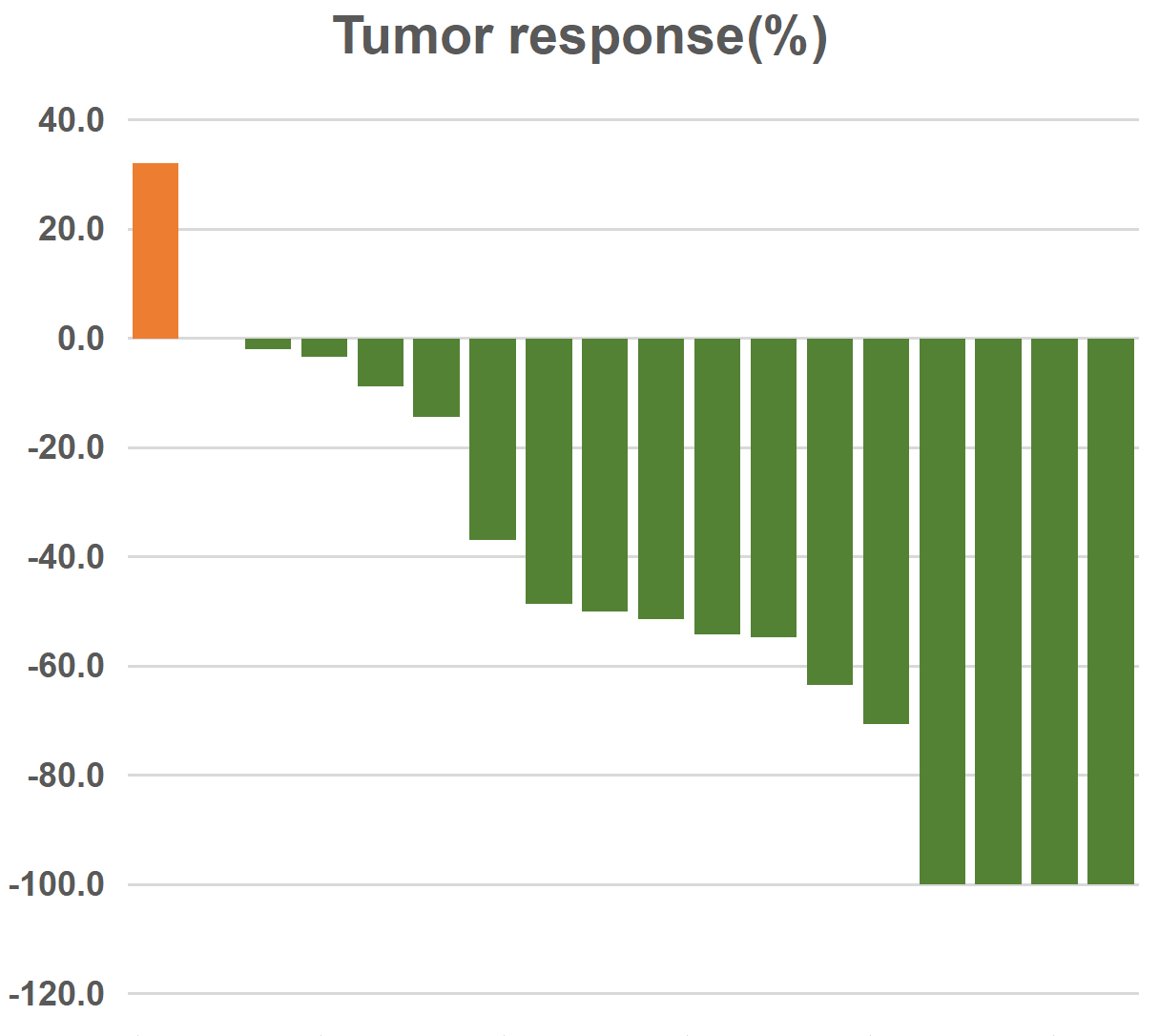
Results: Between July 2021 and October 2022, 21 patients were enrolled. At this preliminary analysis (data cutoff, October, 2022), median follow-up was 9 months (0.9-15.2 months). Nineteen patients were available for efficacy assessment. Confirmed complete response rate was 15.8% (3/19), ORR was 63.1% (12/19). Disease-controlled rate (DCR) was 89.4% (17/19). The median of progression-free survival (PFS) was not reached, with a high 12-month PFS rate of 72.3%. All grade and = 3 treatment-emergent adverse events occurred in 95% (20/21) and 23.8% (5/21), respectively.
Conclusions: Sintilimab in combination with Axitinib had a manageable safety profile and achieved a promising tumor response rate in patients with advanced FH-RCC. The trial is an ongoing study, with a total of planned 41 patients from 8 sites. The study start date was June 2, 2021. SOURCE OF
Funding: This work was supported by the Natural Science Foundation of China (NSFC 82172785, 82103097, 81902577, 81974398, 81872107 and 81872108), China Postdoctoral Science Foundation (2020M673239, 2021M692286 and 2021M692281), Research Foundation for the Postdoctoral Program of Sichuan University (2021SCU12014 and 2022SCU12042), 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYJC21020), and Science and Technology Support Program of Sichuan Province (2021YFS0119), Post-Doctor Research Project, West China Hospital, Sichuan University (20HXBH026, 2021HXBH036 and 2021HXBH028).
Podium Session
Session: PD24: Kidney Cancer: Advanced (including Drug Therapy) II
PD24-09: Phase II, Multi-center Study of Sintilimab in Combination with Axitinib in Patients with Advanced Fumarate Hydratase-deficient Renal Cell Carcinoma
Sunday, April 30, 2023
8:20 AM – 8:30 AM CST
Location: S401C

Xingming Zhang
West China Hospital, Sichuan University
Podium Presenter(s)
Introduction: Fumarate hydratase-deficient renal cell carcinoma (FH-RCC) is a rare and highly aggressive cancer mainly caused by germline or somatic aberrant of FH gene. Unfortunately, there are no standard treatment. Here we report the preliminary results of a phase II study investigating the efficacy and safety of sintilimab in combination with axitinib in patients with FH-RCC.
Methods: Patients were treated at mutli-center in hospitals of the republic of China. Eligibility criteria included age = 18 years and newly diagnosed as FH-RCC by FH immunohistochemistry and next-generation sequencing or multiplex ligation-dependent probe amplification. Patients received sintilimab (intravenous injection, every 3 week) in combination with axitinib (5mg, orally taken per day) as first-line treatment until disease progression or intolerant to treatment. The primary end point was objective response rate (ORR; RECIST v1.1). This study is registered with ClinicalTrials.gov, NCT04387500.
Results: Between July 2021 and October 2022, 21 patients were enrolled. At this preliminary analysis (data cutoff, October, 2022), median follow-up was 9 months (0.9-15.2 months). Nineteen patients were available for efficacy assessment. Confirmed complete response rate was 15.8% (3/19), ORR was 63.1% (12/19). Disease-controlled rate (DCR) was 89.4% (17/19). The median of progression-free survival (PFS) was not reached, with a high 12-month PFS rate of 72.3%. All grade and = 3 treatment-emergent adverse events occurred in 95% (20/21) and 23.8% (5/21), respectively.
Conclusions: Sintilimab in combination with Axitinib had a manageable safety profile and achieved a promising tumor response rate in patients with advanced FH-RCC. The trial is an ongoing study, with a total of planned 41 patients from 8 sites. The study start date was June 2, 2021. SOURCE OF
Funding: This work was supported by the Natural Science Foundation of China (NSFC 82172785, 82103097, 81902577, 81974398, 81872107 and 81872108), China Postdoctoral Science Foundation (2020M673239, 2021M692286 and 2021M692281), Research Foundation for the Postdoctoral Program of Sichuan University (2021SCU12014 and 2022SCU12042), 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYJC21020), and Science and Technology Support Program of Sichuan Province (2021YFS0119), Post-Doctor Research Project, West China Hospital, Sichuan University (20HXBH026, 2021HXBH036 and 2021HXBH028).


