Laboratory Research
(LR-028) A novel anti-biofilm enzymatic wound gel for treatment of chronic wounds
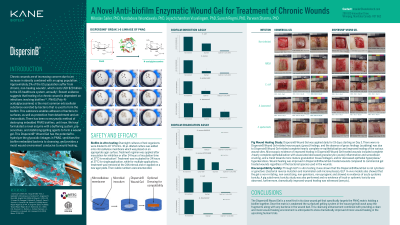
Biofilm is present in over 60% of chronic wounds, contributing to prolonged treatment times, higher risk of reinfection, and increased resistance to antimicrobial treatments. These biofilms are imbedded deep in chronic wound tissue and often contain PNAG (poly-beta 1,6 N-acetyl glucosamine) as the structural binding substance; there is no method of destroying imbedded PNAG biofilms, until now. We have formulated a novel enzyme with a buffering system, preservatives, and stabilizing/gelling agents to form a wound gel. This innovative wound gel (IWG) has the potential to hydrolyse the glycosidic linkages in PNAG and sensitizes the biofilm embedded bacteria to antimicrobial treatments.
Methods:
By isolating and sequencing the code used we have made clones and have expressed and isolated the novel recombinant protein – DispersinB. In vitro efficacy of the IWG and a PHMB containing wound dressing combination was tested for anti-biofilm activity against Staphylococcus epidermidis, Staphylococcus aureus, Acetobacter baumannii, and Escherichia col using a biofilm inhibition and eradication assay. The IWG was tested for safety as per ISO 10933. A porcine wound healing study was performed using wounds infected with the same organisms used for in vitro testing. In addition, shelf-life testing was performed to assess stability of enzyme in the formulation.
Results:
In-vitro data show IWG with a PHMB dressings have a synergistic effect in biofilm inhibition for all organisms tested causing ≥ 5 log CFU reduction in viable numbers of test organisms. When pre-formed biofilms of these organisms were tested, sequential application of IWG with a PHMB dressing daily for 3 days caused ≥ 7 log CFU reduction in viable numbers of test organisms. Biocompatibility studies showed that IWG was non-cytotoxic, non-genotoxic, non-irritating, non-sensitizing, non-toxic, and non-pyrogenic. Porcine wound healing study showed that IWG higher healing rates than control gel. Enzyme was stable in the formulation for more than a year.
Discussion:
The IWG has a novel recombinant enzyme containing product that breaks down the protective PNAG biofilm barrier within a matter of minutes of application and sensitizes the bacteria to antimicrobial treatments In-vitro data demonstrated that there was a synergistic effect when IWG was used with an antimicrobial dressing. Biocompatibility studies performed as per ISO 10933 guidelines and porcine wound healing study showed formulation was biocompatible and significantly improved wound healing. With demonstrated shelf-life of the IWG for over a year. safety, and efficacy, the IWG has the potential to become a key product in the arsenal for treating chronic wounds.
Trademarked Items: DispersinB
References:

.png)