Laboratory Research
(LR-026) Biological Evaluation of a Novel Lyophilized Amniotic Membrane Allograft
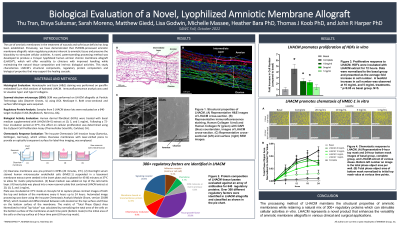
The potential of human amniotic membranes in wound healing and soft tissue repair has long been established. Previous studies have demonstrated that amniotic membrane allografts retain regulatory proteins inherent to amniotic tissue and preserve the bioactivity to stimulate cellular activities. A novel, patent-pending processing method was developed to produce a tri-layer lyophilized human amnion chorion membrane allograft (LHACM*), which will offer versatility to clinicians with improved handling while maintaining the natural tissue composition and intrinsic biological activities. This study characterizes LHACM’s structural components, regulatory protein composition and biological properties that may support the healing cascade.
Methods:
LHACM was prepared using a process modified to accommodate the retention of the intermediate layer. The structure of LHACM was visualized with hematoxylin and eosin (H&E) staining and scanning electron microscopy (SEM). Immunofluorescence analysis was used to identify matrix components, type I and type IV collagens. The regulatory protein content was analyzed using a multiplex ELISA system (RayBiotech). Cellular responses were assessed using multiple in vitro assays. The proliferative effect was quantified on human dermal fibroblasts (HDF) treated with LHACM eluates. The chemotactic response to LHACM was evaluated by a transmembrane assay using human microvascular endothelial cells (HMEC-1).
Results:
Histological evaluation and SEM of LHACM visualized the lamination of the three layers (amnion, intermediate layer, and chorion), indicating that these layers were preserved throughout tissue processing. Immunofluorescence microscopy showed the distribution of matrix components type I and type IV collagens. Screening of regulatory proteins identified a diverse array of bioactive proteins including growth factors, cytokines, chemokines, and protease inhibitors, retained in LHACM. In vitro treatment of primary cells with LHACM eluates induced a proliferative response in HDFs and promoted chemotactic invasion of HMEC-1 cells toward the chamber containing the LHACM eluate.
Discussion:
The processing method of LHACM maintains the structural properties of amniotic membranes while retaining a natural mix of regulatory proteins which can stimulate cellular activities in vitro. LHACM represents a novel product that enhances the versatility of amniotic membrane allografts in various clinical and surgical applications.
Trademarked Items: *AMNIOEFFECT, MIMEDX Group Inc. Marietta, GA
References:

.png)