Practice Innovations
(PI-005) Re-Defining Wound Healing in Chronic Diabetic Foot Ulcers Utilizing Near-Infrared Spectroscopy (NIRS)
A healed wound is the definition of success and a primary study end-point in wound care, however there is no standardized definition. Federal guidance recommends defining complete wound closure as “skin reepithelialization without drainage or dressing requirements confirmed at two consecutive study visits 2 weeks apart”. Despite this recommendation, the definition of a healed wound in high-level clinical studies varies significantly.
While a healed wound is a critical clinical and study end-point signifying successful intervention, failure to maintain a healed wound remains high.
The ability to have an objective point-of-care measurement indicating full-thickness healing could potentially decrease the recurrence rate.
Methods:
To evaluate if point-of-care near infrared spectroscopy (NIRS) can be used as an objective measure for wound healing, thirteen patients in an outpatient wound care clinic were followed with serial NIRS studies. Full-thickness healing was defined as the resolution of deep tissue inflammation as measured by NIRS. The time between epithelialization and resolution of inflammation, as measured by NIRS, was recorded.
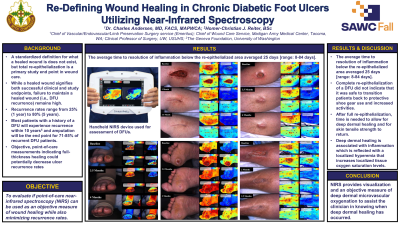
Results:
The average time to see resolution of the inflammation below the area of epithelization averaged 25 days with a range of 8 to 84 days.
Discussion:
Complete epithelization of a DFU is not a good indicator that it is safe to transition a patient back to protective shoe gear use and increased activities. Time is needed to allow for deep dermal healing and increase in skin tensile strength to occur once a wound is fully re-epithelialized.
Deep dermal healing, occurring after reepithelization, is associated with inflammation and associated increase tissue oxygenation saturation levels. This study demonstrates how NIRS may be used to provide visualization and an objective measure of deep dermal microvascular oxygenation to assist the clinician in knowing when deep dermal healing occurred
Trademarked Items:
References: 1. U.S. Department of Health and Human Services, Food and Drug Administration Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER), Center for Devices and Radiological Health (CDRH). Guidance for Industry Chronic Cutaneous Ulcer and Burn Wounds — Developing Products for Treatment. June 2006.
2. Snyder D, Sullivan N, Margolis D, Schoelles K. Skin Substitutes for Treating Chronic Wounds. Technology Assessment Program – Technical Brief. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb 2.
3. Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med. 2017;376(24):2367-2375.
4. López-Moral M, Lázaro-Martínez JL, García-Morales E, García-Álvarez Y, Álvaro-Afonso FJ, Molines-Barroso RJ. Clinical efficacy of therapeutic footwear with a rigid rocker sole in the prevention of recurrence in patients with diabetes mellitus and diabetic polineuropathy: A randomized clinical trial. PLoS One. 2019;14(7):e0219537.
5. Vedhara K, Beattie A, Metcalfe C, et al. Development and preliminary evaluation of a psychosocial intervention for modifying psychosocial risk factors associated with foot re-ulceration in diabetes. Behav Res Ther. 2012;50(5):323-332.
6. Shahin TB, Vaishnav KV, Watchman M, et al. Tissue augmentation with allograft adipose matrix for the diabetic foot in rem Surg Glob Open. 2017;5(10):e1555.
7. Nickerson DS. Reconsidering nerve decompression: an overlooked opportunity to limit diabetic foot ulcer recurrence and amputation. J Diabetes Sci Technol. 2013;7(5):1195-1201.
8. Najafi B, Reeves ND, Armstrong DG. Leveraging smart technologies to improve the management of diabetic foot ulcers and extend ulcer-free days in remission. Diabetes Metab Res Rev. 2020;36(Suppl 1):e3239.

.png)