Laboratory Research
(LR-007) Careful formulation techniques can ensure that pH controlled hypochlorous acid (pHA) wound cleansers remain stable in a tightly controlled pH range over its entire shelf life.
Pure Hypochlorous Acid (pHA) based cleansers are recommended for use by several important guidelines. Much clinical research supports the concept of using pHA as a safe and effective method to cleanse wounds of germs, biofilm, and debris. A pHA solution remains pure only between pH 3.5 to 5.5. Change from this range on the lower side will create toxic chlorine gas. On the higher side, highly cytotoxic hypochlorite ion (bleach) will begin to form. Hypochlorous acid in solution is inherently unstable, and special formulation and packaging techniques are essential to retain a pHA solution in this range over many months.
Methods:
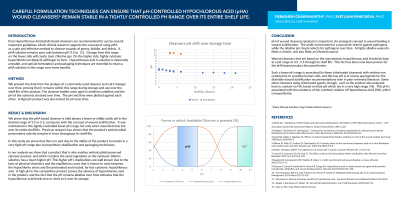
We present the data from the analysis of a commonly used cleanser as its pH changes over time, proving that it remains within this range during storage and use over the shelf life of the product. The cleanser bottles were aged in ambient condition and the pH of the contents checked over time. The pH and time were plotted against each other. A high pH product was also tested for pH over time.
Results:
We prove that the pHA based cleanser is held always a lower pH in the desired range of 3.5 to 5.5, conducive with the concept of wound acidification. It maintained in this tightly controlled lower pH range not only when manufactured, but over its entire shelf life. We show that the competitive product was of a much higher pH and thus known to contain not hypochlorous acid (HA), but hypochlorite based on laws of physical chemistry related to the chemical equilibrium between the desired hypochlorous acid and the undesirable hypochlorite.
Discussion:
pH of wound cleansers/products is important. An emergent concept in wound healing is wound acidification. Wound cleansers that based on the noncytotoxic hypochlorous acid should be kept at a pH range of 3.5 -5.5 through its shelf life. This has been now proven for pHA. pHA therefore is non cytotoxic to sensitive human cells, and additionally, appropriate for the desirable wound acidification. Some other cleansers using chlorinated agents, though, contain no HA, based on their pH which are in a very high range ( >8). This pH is associated with the cytotoxic relative of HA, which is hypochlorite (bleach).
Trademarked Items: Vashe Wound Solution
References: 1. Armstrong DG, Bohn G, Glat P, Kavros SJ, Kirsner R, Snyder R, Tettelbach W.Armstrong DG, et al. Expert Recommendations for the Use of Hypochlorous Solution: Science and Clinical Application. Ostomy Wound Management 2015 May;61(5): S2-S19. https://pubmed.ncbi.nlm.nih.gov/28692424/
2. Block, Michael S, and Brian G Rowan. “Hypochlorous Acid: A Review.” Journal of oral and maxillofacial surgery: official journal of the American Association of Oral and Maxillofacial Surgeons vol. 78,9 (2020): 1461-1466. https://europepmc.org/article/med/32653307
3. Dissemond J. Wound cleansing: benefits of hypochlorous acid. Journal of Wound Care Published Online:5 Oct 2020https://www.karger.com/Article/Abstract/481545
4.. Gold MH, Andriessen A, Bhatia AC, Bitter P Jr, Chilukuri S, Cohen JL, Robb CW. Topical stabilized hypochlorous acid: The future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020 Feb;19(2):270-277. https://pubmed.ncbi.nlm.nih.gov/31904191/

.png)