Laboratory Research
(LR-020) Characterization of a Novel, Lyophilized Amniotic Membrane Allograft
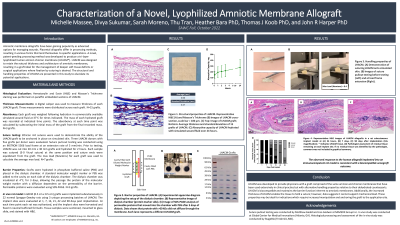
Amniotic membrane allografts have been gaining popularity as advanced options for managing wounds. Placental allografts differ in processing methods, resulting in various forms that lend themselves to specific applications. A novel, patent-pending processing method was developed to produce a, tri-layer lyophilized human amnion chorion membrane (LHACM*). LHACM was designed to retain the natural thickness and architecture of amniotic membranes, resulting in a graft ideal for the management of deeper soft tissue deficits or surgical applications where fixation by suturing is desired. The structural and handling properties of LHACM are presented in this study to elucidate its potential applications.
Methods:
LHACM is prepared using the PURION process with modifications to include the intermediate layer. Thickness of dry grafts was measured using calipers and structure was visualized with H&E staining. Using simulated skin and Ethicon 4-0 sutures, the ability of the graft to be anchored in place was evaluated. Hydration rate and maximum absorbency were determined by exposing the graft to synthetic wound fluid and measuring fluid uptake. The barrier properties were demonstrated using a dialysis chamber and permeable proteins were evaluated using SDS-PAGE. In vivo assessment was performed using a 90-day rat dorsal subcutaneous implant model.
Results:
LHACM grafts’ thickness averaged 0.6 mm ± 0.4 mm. H&E staining confirmed the presence of amnion, intermediate and chorion layers. Simulated-use demonstrated the ability of the graft, in both dry and wet states, to be secured by suturing. The structural integrity of the graft was evident, as it served as a barrier to prevent the diffusion of high molecular weight proteins. However, the hygroscopic nature of the lyophilized graft did allow for uptake of wound fluid into the tissue. LHACM was well-tolerated when implanted in a normal rat, demonstrating biocompatibility and initiation of bioresorption during the 90-day study.
Discussion: LHACM was developed to provide physicians a graft comprised of the same amnion and chorion membranes which have been used extensively in clinical practice, but with alternative handling properties relative to dehydrated amnion/chorion grafts. The processing methods resulted in a graft that more closely retained the thickness and handling of the natural tissue. In vitro and in vivo testing confirmed these process changes did not compromise the graft’s intended use as a barrier, nor the biocompatibility profile.
Trademarked Items: *AMNIOEFFECT, MIMEDX Group Inc. Marietta, GA
References:

.png)