Back
Clinical Research
(CR-002) Point-of-care fluorescence imaging reveals extent of chronic inhibitory bacterial load in diabetic foot ulcers

Co-Author(s):
Michael Edmonds, MD; Monique Rennie, PhD; Thomas Serena, MD, FACS, MAPWCA
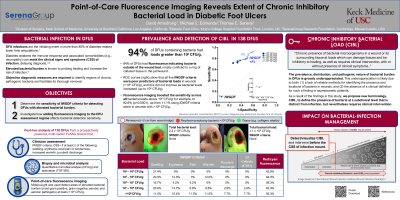
<b>Introduction</b>: <p class="MsoNormal"><span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">Elevated levels of bacteria, including biofilm, increase the risk of infection and its complications as well as hamper healing in diabetic foot ulcers (DFUs). The challenge of addressing high bacterial load in the absence of clinical signs and symptoms of infection (CSS) is exacerbated by a lack of clinical terminology and diagnostic tools. The present study aims to depict the role of fluorescence imaging as a point-of-care, non-invasive solution to detect biofilm encased and planktonic bacteria at high loads in wounds without the need to rely on CSS. </span></p><br/><br/><b>Methods</b>: <p class="MsoNormal"><span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">A post-hoc analysis of a multicenter (14 sites across the Unites States) clinical trial was conducted. </span>Sensitivity and specificity of International Wound Infection Institute (IWII) 2016 clinical signs and symptoms (CSS) of infection criteria and point-of-care imaging of bacterial fluorescence were obtained and compared for detection across bacterial loads of concern (10<sup>4 </sup>CFU/g to 10<sup>9</sup> CFU/g) in 138 DFUs. Quantitative biopsy analysis confirmed total bacterial load.<span style="mso-spacerun: yes;"> </span></p><br/><br/><b>Results</b>: <p class="MsoNormal"><span lang="EN-US" style="mso-ansi-language: EN-US;">131 DFUs had confirmed bacterial presence, of which </span>93%(123/131) had bacterial loads >10<sup>4</sup>CFU/g<span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">. In those wounds, CSS were largely absent, and no better than chance in detecting high bacterial load presence. ROC curves showed that detection of CSS </span>did not correlate to, or increase proportionately with, high bacterial loads (10<sup>4</sup>CFU/g to 10<sup>9</sup>CFU/g)<span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">; CSS prevalence was < 12% in DFUs with >10<sup>8</sup>CFU/g. Fluorescence imaging increased bacterial detection sensitivity across loads 10<sup>4</sup> to 10<sup>9</sup> (p < 0.0001); sensitivity of fluorescence peaked at >10<sup>8</sup>CFU/g (92.6% vs. 11.1% for CSS). Imaging revealed that the periwound had elevated bacterial loads in 84.2% of the 138 DFUs. <span style="mso-spacerun: yes;"> </span></span></p><br/><br/><b>Discussion</b>: <p class="MsoNormal"><span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">A new clinical terminology is introduced: <strong>chronic inhibitory bacterial load (CIBL)</strong>, as a biomarker to diagnose this CSS-independent, pre-infection state in the diabetic ulcer and periwound skin. </span>We anticipate that the definition of CIBL will spark a paradigm shift in DFU wound assessment and management, which encourages and enables earlier intervention along the bacterial-infection continuum <span style="mso-bidi-font-family: Calibri; mso-bidi-theme-font: minor-latin;">–</span> thereby preventing the sequelae of infection and supporting improved DFU outcomes. Fluorescence imaging has enormous potential in facilitating early bacterial intervention, monitoring treatment effectiveness during and after debridement, aiding antimicrobial stewardship to limit antibiotic and antimicrobial dressing prescriptions, and to improve wound healing outcomes.</p><br/><br/><b>Trademarked Items</b>: <br/><br/><b>References</b>: <br/><br/>

.png)