Back
Case Series/Study
(CS-048) Negative Pressure Wound Therapy with Instillation and Silicone-Acrylate Hybrid Drape Use in Complex Wounds: A Small Case Series

Co-Author(s):
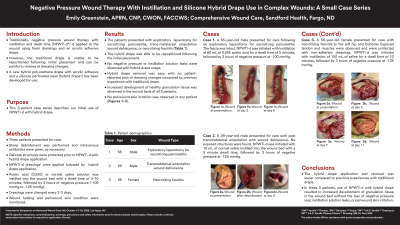
<b>Introduction</b>: <span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;">Negative pressure wound therapy with instillation and dwell time (NPWTi-d*) using a reticulated open cell foam dressing with through holes (ROCF-CC<sup>†</sup>) has been reported to help solubilize non-viable tissue and thick exudate as well as remove wound debris and infectious materials.</span><span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;"><sup><span style="mso-no-proof: yes;">1-3</span></sup></span><span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;"> NPWTi-d has traditionally used an adhesive acrylic drape. A hybrid polyurethane drape with acrylic adhesive and silicone perforated layer (hybrid drape<sup>‡</sup>) has been developed to be easy to apply and remove and be repositioned upon first application. The hybrid drape has been associated with reduced periwound skin irritation and patient pain upon dressing changes.</span><span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;"><sup><span style="mso-no-proof: yes;">4,5 </span></sup></span><br/><br/><b>Methods</b>: <span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;">The use of NPWTi-d with ROCF-CC and hybrid drape was assessed in 3 patients with complex wounds. Patients and wounds were assessed at presentation. Antibiotics were initiated and surgical debridement performed as needed. Delicate structures were protected with a non-adherent hydrofiber dressing. NPWTi-d with ROCF-CC and hybrid drape were applied to the wounds. Normal saline or acetic acid (0.25%) was instilled with a dwell time of 10 minutes, followed by continuous negative pressure at -125 mmHg for 2 to 3.5 hours. Dressing changes occurred every 2-3 days. NPWTi-d was discontinued once healthy granulation tissue covered the wound bed.</span><br/><br/><b>Results</b>: <p class="MsoNormal"><span style="font-family: 'Arial',sans-serif;">Three patients (age range 48-78 years) presented for care. Previous medical history included paraplegia, venous insufficiency, and poor wound healing. Wound types managed included bilateral decubitus ulcers, venous leg ulcer, and an above the knee amputation. One patient received normal saline while the other two received acetic acid (0.25%) for instillation solution. No instillation or negative pressure leaks were observed with the use of hybrid drape. NPWTi-d was discontinued in all patients after granulation tissue completely covered the wound bed. Previous periwound skin irritation and wound edges were improved. No further irritation was noted with the use of hybrid drape. </span></p><br/><br/><b>Discussion</b>: <span style="font-size: 11.0pt; line-height: 107%; font-family: 'Arial',sans-serif; mso-fareast-font-family: Calibri; mso-fareast-theme-font: minor-latin; mso-ansi-language: EN-US; mso-fareast-language: EN-US; mso-bidi-language: AR-SA;">Use of NPWTi-d with ROCF-CC and hybrid drape resulted in the development of healthy granulation tissue in the wound bed without periwound skin irritation or instillation or negative pressure leaks for all 3 patients.</span><br/><br/><b>Trademarked Items</b>: *3M™ Veraflo™ Therapy, †3M™ V.A.C. Veraflo Cleanse Choice™ Dressing, ‡3M™ Dermatac™ Drape (3M Company, San Antonio, TX)<br/><br/><b>References</b>: therapy with instillation to remove thick exudate. Int Wound J. 2/28/2017 2017;14(5):842-848. doi:10.1111/iwj.12719
2. Fernandez LG, Matthews MR, Ellman C, Jackson P, Villareal DH, Norwood S. Use of reticulated open cell foam dressings with through holes during negative pressure wound therapy with instillaton and dwell time: A large case study. Wounds. 2020;32(10):279-282.
3. Blalock L. Use of Negative Pressure Wound Therapy With Instillation and a Novel Reticulated Open-cell Foam Dressing With Through Holes at a Level 2 Trauma Center. Wounds. 2019;31(2):55-58.
4. Fernandez LG, Matthews MR, Benton C, et al. Use of a novel silicone-acrylic drape with negative pressure wound therapy in anatomically challenging wounds. Int Wound J. 2020;17(6):1829-1834. doi:10.1111/iwj.13471
5. Greenstein E, Moore N. Use of a Novel Silicone-Acrylic Drape With Negative Pressure Wound Therapy in Four Patients With Periwound Skin Breakdown. Wounds. 2021;33(11):304-307.<br/><br/>
2. Fernandez LG, Matthews MR, Ellman C, Jackson P, Villareal DH, Norwood S. Use of reticulated open cell foam dressings with through holes during negative pressure wound therapy with instillaton and dwell time: A large case study. Wounds. 2020;32(10):279-282.
3. Blalock L. Use of Negative Pressure Wound Therapy With Instillation and a Novel Reticulated Open-cell Foam Dressing With Through Holes at a Level 2 Trauma Center. Wounds. 2019;31(2):55-58.
4. Fernandez LG, Matthews MR, Benton C, et al. Use of a novel silicone-acrylic drape with negative pressure wound therapy in anatomically challenging wounds. Int Wound J. 2020;17(6):1829-1834. doi:10.1111/iwj.13471
5. Greenstein E, Moore N. Use of a Novel Silicone-Acrylic Drape With Negative Pressure Wound Therapy in Four Patients With Periwound Skin Breakdown. Wounds. 2021;33(11):304-307.<br/><br/>

.png)
