Laboratory Research
(LR-025) Bacterial Susceptibility and Feasibility of Bacterial Clearance in Polyurethane* vs Collagen-Based Dermal Substitutes
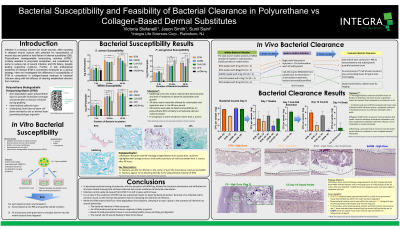
Infection is a serious concern for acute wounds and burns, often resulting in delayed wound closure with potential for exacerbation of wound size and partial or total failure of dermal substitutes (DS). Polyurethane matrices (PUM)*, innately resistant to enzymatic breakdown, are considered by some to reduce risk of wound infection and DS failure, despite lacking supportive evidence. Further, in situ antibacterial treatment of infected PUM is sometimes employed as a clinical strategy. Here we investigated the difference in susceptibility of PUM in comparison to collagen-based matrices to bacterial infection along with the efficacy of clearing established infection from matrices.
Methods:
Susceptibility of PUM versus four collagen-based matrices (urinary bladder, engineered-collagen, fish skin, bovine skin) was explored in vitro with S. aureus, P. aeruginosa, and MRSA. Matrices were inoculated with 103 CFU for each bacterial strain and incubated 2, 24, or 48 hours. Feasibility of bacterial clearance was explored via rodent subcutaneous model wherein DS were pre-loaded with 106 CFU/mL or 109 CFU/mL MRSA, implanted, incubated for 1-week, and subsequently treated, or not, with 2% chlorhexidine irrigation of the subcutaneous pocket plus daily vancomycin injections for 1-week. In both experiments, bacterial counts were obtained via extraction of infected articles in PBS.
Results:
Susceptibility data demonstrated that PUM matrices can sustain similar or greater bacterial loads compared to collagen-based DS across all strains studied. In the rodent subcutaneous model, for all PUM, antibacterial treatment was effective in reducing bacterial load at the lower inoculation level (106 CFU/mL) but not at higher inoculation (109 CFU/mL), with no sample group exhibiting complete clearance at 1 week. Only removal of infected DS from the subcutaneous pocket along with antibacterial treatment allowed for complete infection clearance.
Discussion:
Our data shows that neither PUM or collagen-based DS possess innate resistance to bacterial colonization or infection for the bacterial strains tested. Furthermore, infection cannot be easily eliminated from infected PUM that remains implanted, especially at higher bacterial loads. These results suggest that PUM does not significantly reduce risks of wound infection. For patients at high-risk of infections, clinicians should continue to rely on established risk reduction practices regardless of the chosen DS.
Trademarked Items: *NovoSorb BTM, PolyNovo Biomaterials Pty Ltd., Port Melbourne, Australia
References: Bowler, P. G., Duerden, B. I., & Armstrong, D. G. (2001). Wound microbiology and associated approaches to wound management. Clinical Microbiology Reviews, 14(2), 244–269. https://doi.org/10.1128/CMR.14.2.244-269.2001
Sibbald, R. G., Woo, K., & Ayello, E. A. (2006). Increased bacterial burden and infection: the story of NERDS and STONES. Advances in Skin & Wound Care, 19(8), 443–447. https://doi.org/10.1097/00129334-200610000-00012
Solanki, N. S., York, B., Gao, Y., Baker, P., & Wong She, R. B. (2020). A consecutive case series of defects reconstructed using NovoSorbⓇ Biodegradable Temporising Matrix: Initial experience and early results. Journal of Plastic, Reconstructive & Aesthetic Surgery, 73(10), 1845–1853. https://doi.org/https://doi.org/10.1016/j.bjps.2020.05.067
Wu, S. S., Wells, M., Ascha, M., Gatherwright, J., & Chepla, K. (2022). Performance of biodegradable temporizing matrix vs collagen-chondroitin silicone bilayer dermal regeneration substitutes in soft tissue wound healing: a retrospective analysis. Wounds : A Compendium of Clinical Research and Practice, 34(4), 106–115.

.png)
