Back
Regular Abstract
Real-world Evidence
Session: Poster Session
4: ‘Real world’ impact of immune checkpoint inhibitors (ICI) on survival in metastatic renal cell cancer (mRCC)
Location: Poster Hall, Board A4
Emma Baghurtst, BSc; Timothy Eisen, FRCP, FMedSci; Jessica Clifton, MBBS; Kate Fife, MD, FRANZCR, FRCR, FRCP; Henno Martin , MRCP, FRCR; Athena Matakidou, MRCP, PhD; Sarah Welsh, MRCP, PhD; Brent O'Carrigan, MRCP, PhD; Mary Fortune, PhD

Will Ince, FRCR MRCP MRCGP MB BS
Specialist Registrar
Addenbooke's Hospital
Cambridge, England, United Kingdom
Poster Presenter(s)
Background
There have been rapid changes in the treatment paradigm for patients with mRCC during the last decade. Tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICI) alone and in combination are now approved in first and subsequent line settings. There is a paucity of data about the impact of ICI on survival in ‘real world’ populations with mRCC. We examined an unselected population of mRCC patients to determine whether ICI at any stage of treatment had an effect on overall survival (OS).
Methods
Patients treated at a single academic centre (Addenbrooke’s Hospital, Cambridge, UK) between January 2012 and July 2020 who received ≥1 line of systemic treatment were included. OS was calculated as time from start of first treatment to death; data was analysed using the Kaplan Meier method and a multivariate Cox proportional hazards regression model adjusting for variables in Table 1.
Results
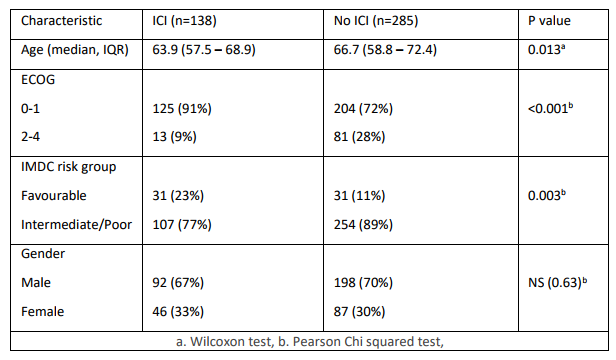
423 patients received ≥1 line of treatment. 285 (67%) received TKI and/or mTOR treatment only and 138 (33%) received ICI at any stage in their treatment. Patients receiving any ICI were more likely to be PS ECOG 0-1 (91% v 72%, p<0.001) and IMDC favourable risk (23% v 11%, p=0.003). On multivariate analysis, ICI at any stage of treatment significantly prolonged overall survival (OS) with a median OS of 34.2 months (95% CI 28.7–46.7) in those treated with ICI compared to 11.3 months (95% CI 9.0–15.1) in those who did not receive ICI (HR 0.45, 95%CI 0.34–0.59, p<0.001). The OS advantage was evident across all IMDC risk strata: favourable risk (HR 0.55, 95%CI 0.21–0.99, p=0.046), intermediate risk (HR 0.51, 95%CI 0.40–0.85, p=0.005) and poor risk subgroups (HR 0.37, 95%CI 0.18–0.54, p<0.001).
Conclusion
Treatment options for mRCC are manifold. Our data suggest that the use of ICI at any line of treatment provides a significant overall survival advantage, controlling for common clinical confounding variables.
There have been rapid changes in the treatment paradigm for patients with mRCC during the last decade. Tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICI) alone and in combination are now approved in first and subsequent line settings. There is a paucity of data about the impact of ICI on survival in ‘real world’ populations with mRCC. We examined an unselected population of mRCC patients to determine whether ICI at any stage of treatment had an effect on overall survival (OS).
Methods
Patients treated at a single academic centre (Addenbrooke’s Hospital, Cambridge, UK) between January 2012 and July 2020 who received ≥1 line of systemic treatment were included. OS was calculated as time from start of first treatment to death; data was analysed using the Kaplan Meier method and a multivariate Cox proportional hazards regression model adjusting for variables in Table 1.
Results
423 patients received ≥1 line of treatment. 285 (67%) received TKI and/or mTOR treatment only and 138 (33%) received ICI at any stage in their treatment. Patients receiving any ICI were more likely to be PS ECOG 0-1 (91% v 72%, p<0.001) and IMDC favourable risk (23% v 11%, p=0.003). On multivariate analysis, ICI at any stage of treatment significantly prolonged overall survival (OS) with a median OS of 34.2 months (95% CI 28.7–46.7) in those treated with ICI compared to 11.3 months (95% CI 9.0–15.1) in those who did not receive ICI (HR 0.45, 95%CI 0.34–0.59, p<0.001). The OS advantage was evident across all IMDC risk strata: favourable risk (HR 0.55, 95%CI 0.21–0.99, p=0.046), intermediate risk (HR 0.51, 95%CI 0.40–0.85, p=0.005) and poor risk subgroups (HR 0.37, 95%CI 0.18–0.54, p<0.001).
Conclusion
Treatment options for mRCC are manifold. Our data suggest that the use of ICI at any line of treatment provides a significant overall survival advantage, controlling for common clinical confounding variables.

