Back
Poster, Podium & Video Sessions
Moderated Poster
MP38: Sexual Function/Dysfunction: Basic Research & Pathophysiology
MP38-01: Bioactive fibrous membrane containing endogenous nerve growth factor promotes cellular and functional recuperation of impaired cavernous nerve: an in vivo animal study
Sunday, May 15, 2022
7:00 AM – 8:15 AM
Location: Room 222
Paulo Mota, Cabeceiras de basto, Portugal, Marta Casanova, Braga, Portugal, Helena Vala, Carmen Nóbrega, Vila Real, Portugal, Alain Morais, Braga, Portugal, Catarina Silva, Guimarães, Portugal, Sara Anacleto*, Braga, Portugal, Alexandre Barros, Guimarães, Portugal, Rui Reis, Braga, Portugal, Estêvão Lima, Cabeceiras de basto, Portugal, Albino Martins, Nuno Neves, Braga, Portugal
- SA
Poster Presenter(s)
Introduction: Prostate cancer is a frequent disease and the radical prostatectomy one of the most used treatment when the cancer is localized. This procedure commonly causes erectile dysfunction (ED) by injure the cavernous nerve (CN) during the surgery.
With this research we investigate the ability of a bioactive fibrous membrane (FM) to regenerate the damaged CN in a rat model of CN injury.
Methods: Male Sprague–Dawley (SD) rats were distributed by 4 experimental groups: sham (only a lower midline abdominal incision), negative control (lower midline abdominal incision with pelvic dissection and CN crush but no FM implantation), electrospun fibrous membrane [eFM] (with CN crush and FM implantation) and electrospun fibrous membrane biofunctionalized with NGF from rat urine [eFM-uNGF] (with CN crush). Function was evaluated five weeks later after an injection of apomorphine by glans observation (visual scoring) and intracavernous pressure (ICP) measurements. Neurogenic genes expression and Histological and Immunohistochemical analysis of CN and cavernous tissue was performed.
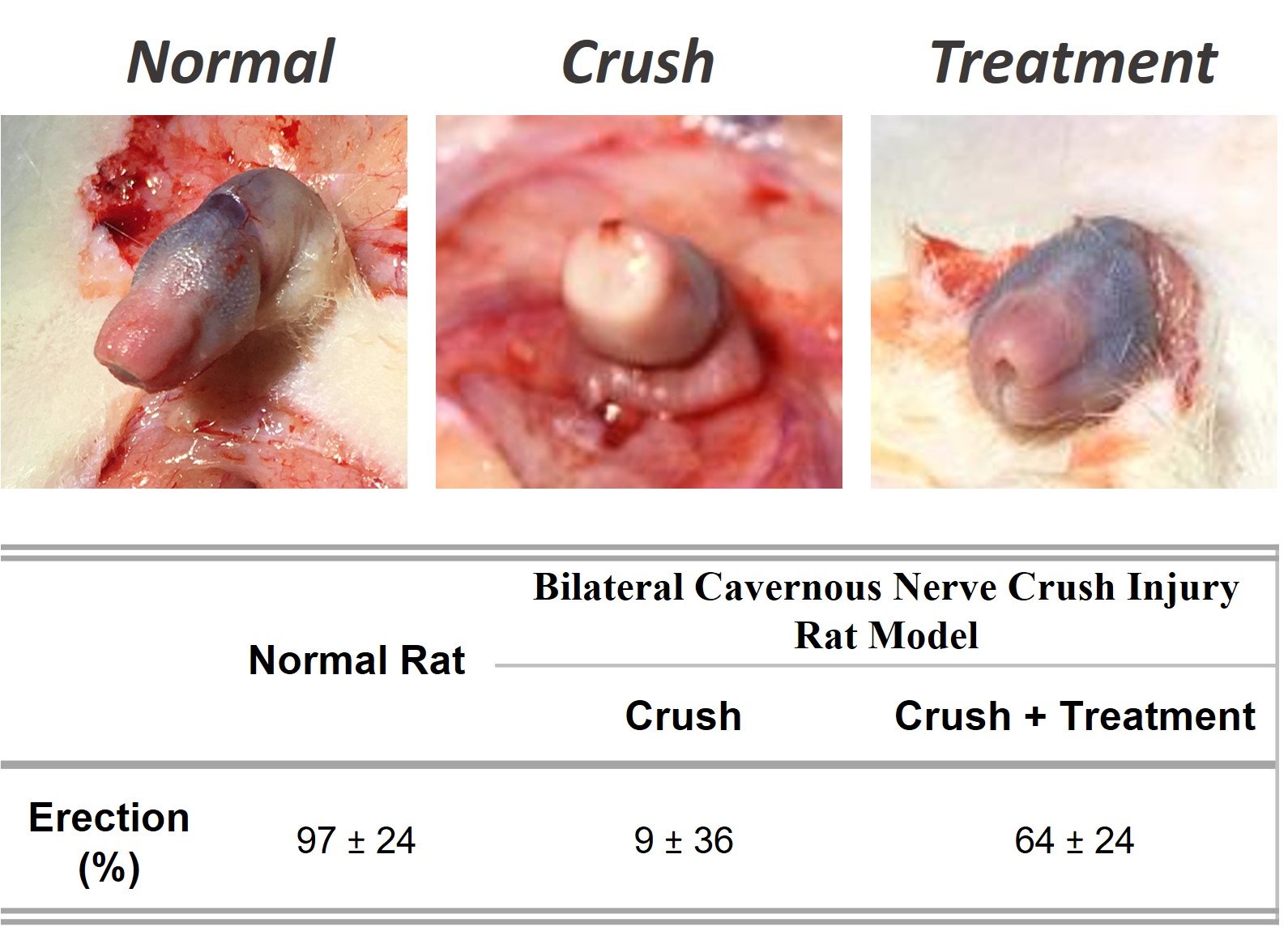
Results: in this model of bilateral CN crush the treatment with implanted bioactive fibrous membrane induces CN regeneration and restoration of erectile function (p < 0.001) (figure), showing a significant increased number of smooth muscle cells and nNOS and eNOS contents. Additionally, the bioactive fibrous membrane promotes a CN protective effect and increase the nerve regeneration capacity by increasing the number of myelinated axons and nNOS-positive cells, recovering from the CN fibrosis observed in rats not treated.
Conclusions: This personalized regenerative strategy could help better recovery of erectile function after CN injuries, and it may constitute an effective novel option to prostate cancer patients suffering from ED after being subject to radical prostatectomy.
Source of Funding: The authors would like to acknowledge the Portuguese Foundation for Science and Technology (FCT) for the PhD grant of M.R.C. (PD/BD/113797/2015) financed by the Doctoral Program on Advanced Therapies for Health (PATH) (FSE/POCH/PD/169/2013), the IF grant of A.M. (IF/00376/2014), the project Cells4_IDs (PTDC/BTM-SAL/28882/2017) and FCT - Portuguese Foundation for Science and Technology, under the project UIDB/04033/2020.
With this research we investigate the ability of a bioactive fibrous membrane (FM) to regenerate the damaged CN in a rat model of CN injury.
Methods: Male Sprague–Dawley (SD) rats were distributed by 4 experimental groups: sham (only a lower midline abdominal incision), negative control (lower midline abdominal incision with pelvic dissection and CN crush but no FM implantation), electrospun fibrous membrane [eFM] (with CN crush and FM implantation) and electrospun fibrous membrane biofunctionalized with NGF from rat urine [eFM-uNGF] (with CN crush). Function was evaluated five weeks later after an injection of apomorphine by glans observation (visual scoring) and intracavernous pressure (ICP) measurements. Neurogenic genes expression and Histological and Immunohistochemical analysis of CN and cavernous tissue was performed.
Results: in this model of bilateral CN crush the treatment with implanted bioactive fibrous membrane induces CN regeneration and restoration of erectile function (p < 0.001) (figure), showing a significant increased number of smooth muscle cells and nNOS and eNOS contents. Additionally, the bioactive fibrous membrane promotes a CN protective effect and increase the nerve regeneration capacity by increasing the number of myelinated axons and nNOS-positive cells, recovering from the CN fibrosis observed in rats not treated.
Conclusions: This personalized regenerative strategy could help better recovery of erectile function after CN injuries, and it may constitute an effective novel option to prostate cancer patients suffering from ED after being subject to radical prostatectomy.
Source of Funding: The authors would like to acknowledge the Portuguese Foundation for Science and Technology (FCT) for the PhD grant of M.R.C. (PD/BD/113797/2015) financed by the Doctoral Program on Advanced Therapies for Health (PATH) (FSE/POCH/PD/169/2013), the IF grant of A.M. (IF/00376/2014), the project Cells4_IDs (PTDC/BTM-SAL/28882/2017) and FCT - Portuguese Foundation for Science and Technology, under the project UIDB/04033/2020.


.jpg)
.jpg)