Back
Poster, Podium & Video Sessions
Late-breaking Abstract I - Cancer & BPH
LBA01-01: Harm-to-Benefit of Three Decades of Prostate Cancer Screening in Black Men
Sunday, May 15, 2022
1:00 PM – 1:10 PM
Location: Room 243
Spyridon Basourakos, Roman Gulati, Randy Vince, Daniel Spratt, Patrick Lewicki, Alexander Hill, Yaw Nyame, Jennifer Cullen, Sarah Markt, Christopher Barbieri, Jim Hu, Jonathan Shoag
- SB
Podium Presenter(s)
Introduction: Prostate-specific antigen screening has profoundly impacted the epidemiology of prostate cancer in the US. Persistent racial disparities in outcomes for Black men warrant re-examination of the harms of screening relative to its cancer-specific mortality benefits in this population.
Methods: We estimated overdiagnoses and overtreatment for all races and Black men aged 50-84 years until 2016, the most recent year with treatment data available, using (a) excess incidence relative to 1986 based on the Surveillance, Epidemiology, and End Results registry and US Census data and (b) an established microsimulation model of prostate cancer natural history. Combining estimates with plausible mortality benefit, we calculated numbers needed to diagnose (NND) and treat (NNT) to prevent one prostate cancer death.
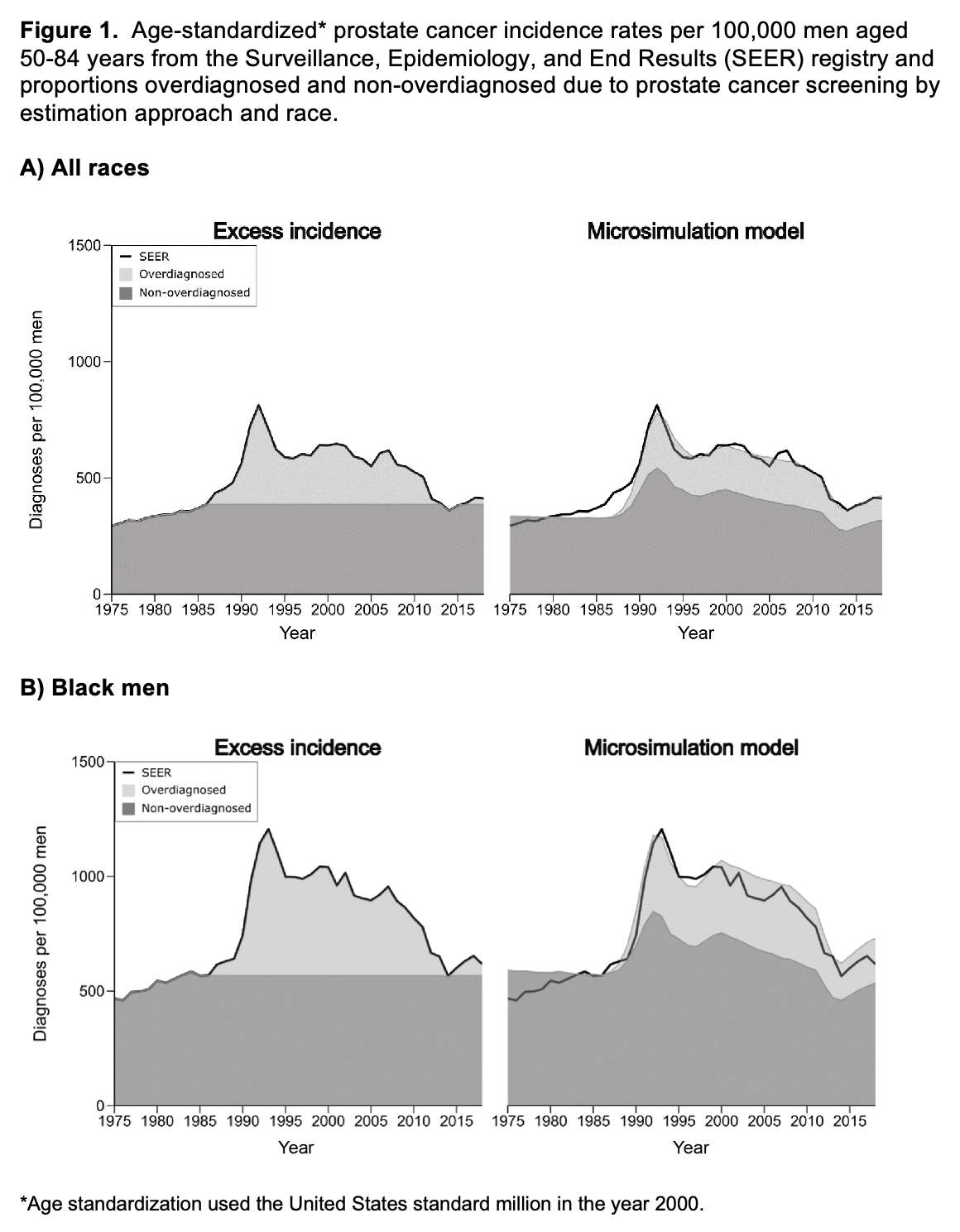
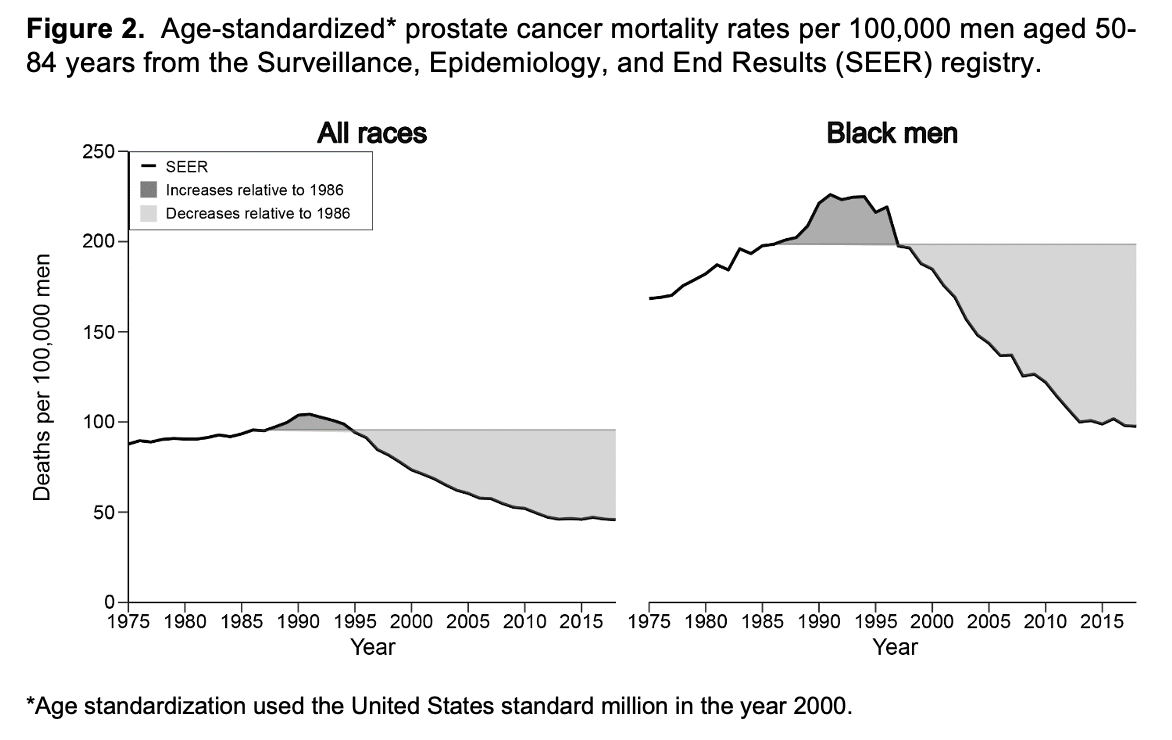
Results: For all races, we estimated 1.5-1.9 million (range between estimation approaches) overdiagnosed and 0.9-1.5 million overtreated prostate cancers by 2016. Figure 1 shows age-standardized prostate cancer incidence partitioned into overdiagnosed and non-overdiagnosed components by estimation approach and race. The two estimation approaches produce similar age-standardized burdens of overdiagnosis over the period 1986-2016. Over this period, prostate cancer mortality initially rose and then declined relative to 1986, with 270,000 fewer deaths among men of all races and 55,000 fewer deaths among Black men (Figure 2). Assuming half of the 270,000 prostate cancer deaths avoided by 2016 were due to screening, the NND and NNT would be 11-14 and 7-11 for all races and 8-12 and 5-9 for Black men. Conservatively accounting for the lag between incidence and mortality, the estimated NND and NNT for black men reach well into the low single digits.
Conclusions: Complementary approaches to quantifying overdiagnosis indicate a harm-benefit tradeoff of prostate-specific antigen screening that is more favorable than previous estimates that used shorter follow-up from the introduction of screening. These data constitute strong support for prostate-specific antigen screening particularly in Black men, for whom there is a paucity of clinical trial evidence.
Source of Funding: JES is supported by the Damon Runyon Research Foundation, Vinney Fund, NIH, and PCORI. JES, SM, and ET are supported by BMS Foundation Cleveland African American Prostate Cancer Project. JCH is supported by the Dow Wallace Fund, NIH and PCORI. RG is supported by NIH. DS reports personal fees from Varian, Janssen, Boston Scientific, AstraZeneca, Blue Earth, Bayer, and funding from Janssen. The remaining authors report no further disclosures.
Methods: We estimated overdiagnoses and overtreatment for all races and Black men aged 50-84 years until 2016, the most recent year with treatment data available, using (a) excess incidence relative to 1986 based on the Surveillance, Epidemiology, and End Results registry and US Census data and (b) an established microsimulation model of prostate cancer natural history. Combining estimates with plausible mortality benefit, we calculated numbers needed to diagnose (NND) and treat (NNT) to prevent one prostate cancer death.
Results: For all races, we estimated 1.5-1.9 million (range between estimation approaches) overdiagnosed and 0.9-1.5 million overtreated prostate cancers by 2016. Figure 1 shows age-standardized prostate cancer incidence partitioned into overdiagnosed and non-overdiagnosed components by estimation approach and race. The two estimation approaches produce similar age-standardized burdens of overdiagnosis over the period 1986-2016. Over this period, prostate cancer mortality initially rose and then declined relative to 1986, with 270,000 fewer deaths among men of all races and 55,000 fewer deaths among Black men (Figure 2). Assuming half of the 270,000 prostate cancer deaths avoided by 2016 were due to screening, the NND and NNT would be 11-14 and 7-11 for all races and 8-12 and 5-9 for Black men. Conservatively accounting for the lag between incidence and mortality, the estimated NND and NNT for black men reach well into the low single digits.
Conclusions: Complementary approaches to quantifying overdiagnosis indicate a harm-benefit tradeoff of prostate-specific antigen screening that is more favorable than previous estimates that used shorter follow-up from the introduction of screening. These data constitute strong support for prostate-specific antigen screening particularly in Black men, for whom there is a paucity of clinical trial evidence.
Source of Funding: JES is supported by the Damon Runyon Research Foundation, Vinney Fund, NIH, and PCORI. JES, SM, and ET are supported by BMS Foundation Cleveland African American Prostate Cancer Project. JCH is supported by the Dow Wallace Fund, NIH and PCORI. RG is supported by NIH. DS reports personal fees from Varian, Janssen, Boston Scientific, AstraZeneca, Blue Earth, Bayer, and funding from Janssen. The remaining authors report no further disclosures.



.jpg)
.jpg)