Back
Poster Session B
Immunobiology
Session: (0578–0588) Cytokines and Cell Trafficking Poster
0587: Increase of Intercellular Adhesion Molecule-1 Linked to Lung Damage of Interstitial Lung Disease in Patients with Autoimmune Diseases
Sunday, November 13, 2022
9:00 AM – 10:30 AM Eastern Time
Location: Virtual Poster Hall
- VP
Veronica Pulito Cueto, PhD
IDIVAL
Santander, Spain
Abstract Poster Presenter(s)
Veronica Pulito-Cueto1, Sara Remuzgo-Martinez1, Fernanda Genre1, Belén Atienza-Mateo1, David Iturbe-Fernández1, Víctor M. Mora-Cuesta1, Leticia Lera-Gómez1, Diana Prieto-Peña1, Virgi Portilla1, Ricardo Blanco2, Oreste Gualillo3, Alfonso Corrales1, José M. Cifrián1, Raquel Lopez Mejias4 and Miguel Ángel González-Gay5, 1Research Group on Genetic Epidemiology and Atherosclerosis in Systemic Diseases and in Metabolic Bone Diseases of the Musculoskeletal System, IDIVAL; and Department of Rheumatology, Hospital Universitario Marqués de Valdecilla, Santander, Spain, 2Hospital Universitario Marqués de Valdecilla, IDIVAL, Santander, Spain, 3Hospital Clínico Universitario de Santiago, SERGAS (Servizo Galego de Saude) and IDIS (Instituto de Investigación Sanitaria de Santiago), NEIRID Lab (Neuroendocrine Interactions in Rheumatology and Inflammatory Diseases), Research Laboratory 9, Santiago de Compostela, Spain, 4IDIVAL, Santander, Spain, 5Department of Medicine and Psychiatry, Universidad de Cantabria; Rheumatology Division, Hospital Universitario Marqués de Valdecilla; Research group on genetic epidemiology and atherosclerosis in systemic diseases and in metabolic diseases of the musculoskeletal system, IDIVAL, Santander, Spain. Cardiovascular Pathophysiology and Genomics Research Unit, School of Physiology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
Background/Purpose: Intercellular adhesion molecule-1 (ICAM-1) regulates leukocyte recruitment from circulation to sites of inflammation, being considered as a marker of underlying endothelial damage [1]. Moreover, ICAM-1 contributes to the development of interstitial lung disease (ILD) and it has been proposed as a prognostic biomarker in idiopathic pulmonary fibrosis (IPF) [2]. Although ILD is one of the main causes of death in patients with autoimmune diseases (AD), mainly in RA and SSc [3], studies on the involvement of ICAM-1 in AD-ILD are scarce. Accordingly, the objective of this work was to elucidate the role of ICAM-1 in the pathological processes of both underlying vascular damage and pulmonary fibrosis in AD-ILD.
Methods: Peripheral venous blood was collected from a total of 144 individuals. Specifically, 57 patients with AD‐ILD+ were recruited: 21 with RA‐ILD+, 21 with SSc‐ILD+ and 15 with other AD‐ILD+. Moreover, we recruited different comparative groups: 45 AD‐ILD− patients (25 RA‐ILD− and 20 SSc‐ILD−); 21 IPF patients; and 21 healthy controls (HC). Serum levels of ICAM-1 were measured by enzyme-linked immunosorbent assay.
Results: Patients with AD-ILD+ exhibited significantly higher ICAM-1 levels than AD-ILD- patients (p< 0.001, Figure 1A). Specifically, RA-ILD+ and SSc-ILD+ patients showed higher levels of ICAM-1 than their ILD- counterparts (p=0.005 and p=0.001, respectively, Figure 1B-C). Furthermore, an increase of ICAM-1 levels was also found in AD-ILD+ patients when compared with HC (p< 0.001, Figure 1A). Likewise, patients with RA-ILD+, SSc-ILD+ and other AD-ILD+ showed higher ICAM-1 levels than HC (p< 0,001, p=0,011 and p=0,002, respectively, Figure 1B-C). However, no difference was observed between AD-ILD+ patients, regardless the underlying AD, and those with IPF (Figure 1A-D).
Conclusion: Our study supports a role of ICAM-1 in the lung damage of AD-ILD+, being the increase of ICAM-1 linked to the presence of ILD in patients with RA and SSc.
References: [1] J Leukoc Biol 2020;108(3):787-799; [2] Eur Respir J 2019;54(3):1900295; [3] Expert Rev Clin Immunol 2018;14(1):69-82.Personal funds, VP-C and SR-M: RD16/0012/0009(ISCIII-ERDF); FG: RICORS Program (RD21/0002/0025) (ISCIII-EU); RL-M: CP16/00033 (ISCIII-ESF).
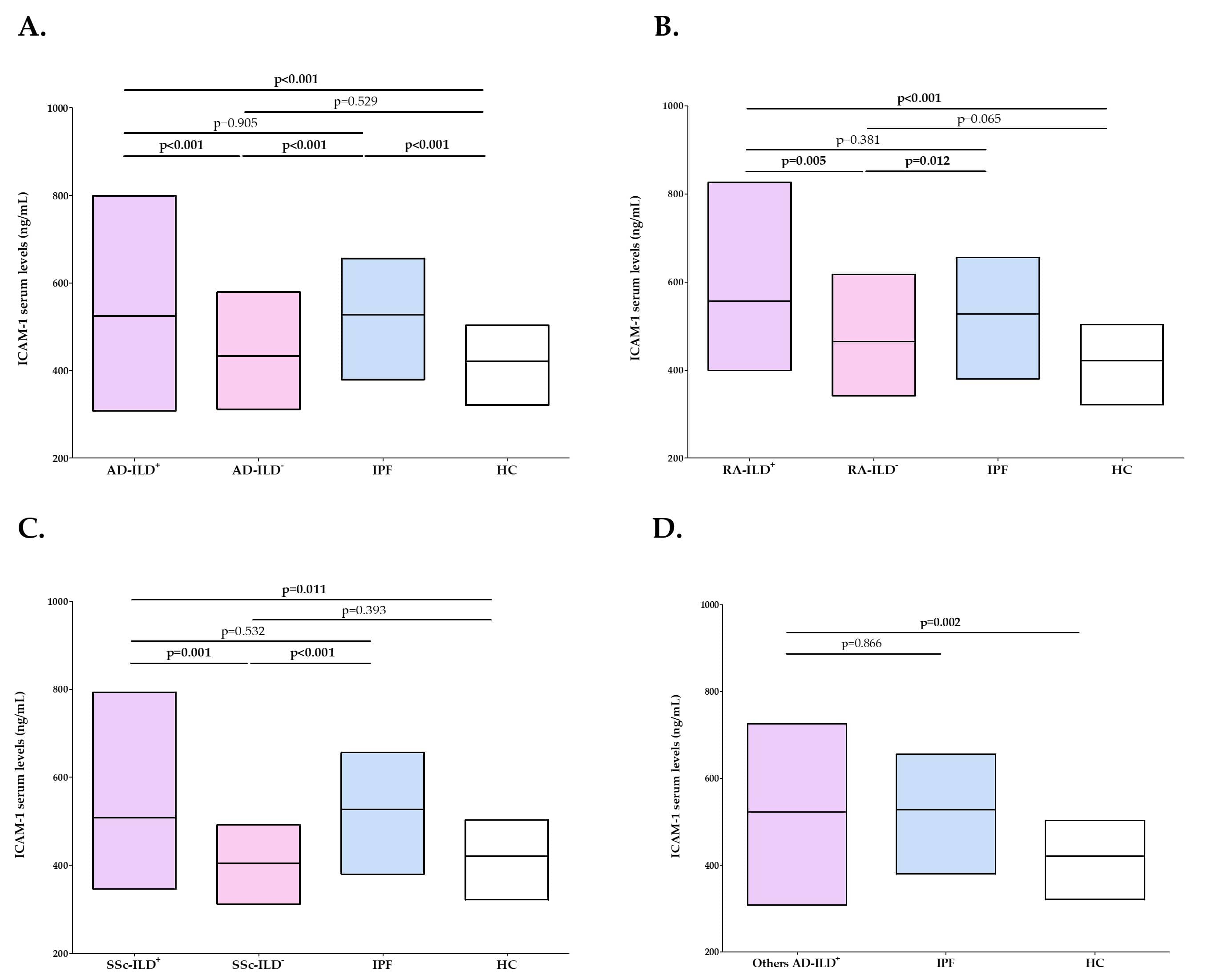 Figure 1. Differences in ICAM-1 serum levels between all the subjects of the study. A. Whole cohort of patients with AD-ILD+ and AD-ILD-, as well as IPF patients and HC; B. Patients with RA-ILD+, RA-ILD- and IPF, as well as HC; C. Patients with SSc-ILD+, SSc-ILD- and IPF, as well as HC; D. Patients with other AD-ILD+ and IPF, as well as HC. Significant results are highlighted in bold.
Figure 1. Differences in ICAM-1 serum levels between all the subjects of the study. A. Whole cohort of patients with AD-ILD+ and AD-ILD-, as well as IPF patients and HC; B. Patients with RA-ILD+, RA-ILD- and IPF, as well as HC; C. Patients with SSc-ILD+, SSc-ILD- and IPF, as well as HC; D. Patients with other AD-ILD+ and IPF, as well as HC. Significant results are highlighted in bold.
Disclosures: V. Pulito-Cueto, None; S. Remuzgo-Martinez, None; F. Genre, None; B. Atienza-Mateo, AbbVie/Abbott, Roche, Pfizer, Celgene, Novartis, Janssen, UCB, Eli Lilly; D. Iturbe-Fernández, None; V. Mora-Cuesta, None; L. Lera-Gómez, None; D. Prieto-Peña, UCB, Roche, Pfizer, Amgen, Janssen, AbbVie/Abbott, Novartis, Eli Lilly; V. Portilla, None; R. Blanco, Eli Lilly, Pfizer, Roche, Janssen, MSD, AbbVie, Amgen, AstraZeneca, Bristol Myers Squibb, Galapagos, Novartis, Sanofi; O. Gualillo, None; A. Corrales, None; J. Cifrián, None; R. Lopez Mejias, None; M. González-Gay, AbbVie/Abbott, Merck/MSD, Janssen, Roche, AbbVie/Abbott, Roche, Sanofi, Eli Lilly, Celgene, Sobi, Merck/MSD.
Background/Purpose: Intercellular adhesion molecule-1 (ICAM-1) regulates leukocyte recruitment from circulation to sites of inflammation, being considered as a marker of underlying endothelial damage [1]. Moreover, ICAM-1 contributes to the development of interstitial lung disease (ILD) and it has been proposed as a prognostic biomarker in idiopathic pulmonary fibrosis (IPF) [2]. Although ILD is one of the main causes of death in patients with autoimmune diseases (AD), mainly in RA and SSc [3], studies on the involvement of ICAM-1 in AD-ILD are scarce. Accordingly, the objective of this work was to elucidate the role of ICAM-1 in the pathological processes of both underlying vascular damage and pulmonary fibrosis in AD-ILD.
Methods: Peripheral venous blood was collected from a total of 144 individuals. Specifically, 57 patients with AD‐ILD+ were recruited: 21 with RA‐ILD+, 21 with SSc‐ILD+ and 15 with other AD‐ILD+. Moreover, we recruited different comparative groups: 45 AD‐ILD− patients (25 RA‐ILD− and 20 SSc‐ILD−); 21 IPF patients; and 21 healthy controls (HC). Serum levels of ICAM-1 were measured by enzyme-linked immunosorbent assay.
Results: Patients with AD-ILD+ exhibited significantly higher ICAM-1 levels than AD-ILD- patients (p< 0.001, Figure 1A). Specifically, RA-ILD+ and SSc-ILD+ patients showed higher levels of ICAM-1 than their ILD- counterparts (p=0.005 and p=0.001, respectively, Figure 1B-C). Furthermore, an increase of ICAM-1 levels was also found in AD-ILD+ patients when compared with HC (p< 0.001, Figure 1A). Likewise, patients with RA-ILD+, SSc-ILD+ and other AD-ILD+ showed higher ICAM-1 levels than HC (p< 0,001, p=0,011 and p=0,002, respectively, Figure 1B-C). However, no difference was observed between AD-ILD+ patients, regardless the underlying AD, and those with IPF (Figure 1A-D).
Conclusion: Our study supports a role of ICAM-1 in the lung damage of AD-ILD+, being the increase of ICAM-1 linked to the presence of ILD in patients with RA and SSc.
References: [1] J Leukoc Biol 2020;108(3):787-799; [2] Eur Respir J 2019;54(3):1900295; [3] Expert Rev Clin Immunol 2018;14(1):69-82.Personal funds, VP-C and SR-M: RD16/0012/0009(ISCIII-ERDF); FG: RICORS Program (RD21/0002/0025) (ISCIII-EU); RL-M: CP16/00033 (ISCIII-ESF).
 Figure 1. Differences in ICAM-1 serum levels between all the subjects of the study. A. Whole cohort of patients with AD-ILD+ and AD-ILD-, as well as IPF patients and HC; B. Patients with RA-ILD+, RA-ILD- and IPF, as well as HC; C. Patients with SSc-ILD+, SSc-ILD- and IPF, as well as HC; D. Patients with other AD-ILD+ and IPF, as well as HC. Significant results are highlighted in bold.
Figure 1. Differences in ICAM-1 serum levels between all the subjects of the study. A. Whole cohort of patients with AD-ILD+ and AD-ILD-, as well as IPF patients and HC; B. Patients with RA-ILD+, RA-ILD- and IPF, as well as HC; C. Patients with SSc-ILD+, SSc-ILD- and IPF, as well as HC; D. Patients with other AD-ILD+ and IPF, as well as HC. Significant results are highlighted in bold.Disclosures: V. Pulito-Cueto, None; S. Remuzgo-Martinez, None; F. Genre, None; B. Atienza-Mateo, AbbVie/Abbott, Roche, Pfizer, Celgene, Novartis, Janssen, UCB, Eli Lilly; D. Iturbe-Fernández, None; V. Mora-Cuesta, None; L. Lera-Gómez, None; D. Prieto-Peña, UCB, Roche, Pfizer, Amgen, Janssen, AbbVie/Abbott, Novartis, Eli Lilly; V. Portilla, None; R. Blanco, Eli Lilly, Pfizer, Roche, Janssen, MSD, AbbVie, Amgen, AstraZeneca, Bristol Myers Squibb, Galapagos, Novartis, Sanofi; O. Gualillo, None; A. Corrales, None; J. Cifrián, None; R. Lopez Mejias, None; M. González-Gay, AbbVie/Abbott, Merck/MSD, Janssen, Roche, AbbVie/Abbott, Roche, Sanofi, Eli Lilly, Celgene, Sobi, Merck/MSD.

